Animal experiments
Porcine intravenous bacteraemia
The experimental protocol for the pig trial was revised and approved by University of Bologna animal welfare body first, and then by the Italian Ministry of Health as dictated by the Legislative Decree 26/2014 implementing the European Directive 2010/63/EU (approval n° 594/2021-PR). All procedures were performed according to relevant guidelines and regulations, and methodologies are reported in accordance with Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.
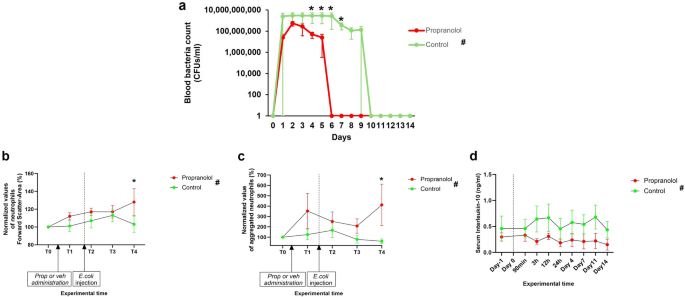
Hybrid breed female pigs (n = 8), weighing 44 ± 7 kg, were divided equally into two groups: propranolol-group and control-group (n = 4 each). Five days (Day -5) before the immune challenge, pigs were implanted, under general anaesthesia, with a surgically-inserted double lumen central venous catheter allowing repeated blood samplings and bacterial infusion (Supplementary methods, page 2). Anaesthesia was achieved with an intramuscular injection of tiletamine-zolazepam (5 mg/kg; Zoletil, Virbac S.r.l., Milan, Italy) followed by an intravenous (i.v.) bolus of propofol (2–4 mg/kg; Proposure, Boehringer Ingelheim Animal Health Italia S.p.a., Noventana, Italy), and maintained with Sevoflurane (2–4% in 1:1 air/oxygen mixture; SevoFlo, Zoetis S.r.l., Rome, Italy) upon oro-tracheal intubation. Animals in the propranolol-group were orally administered 3 mg/kg of DL-Propranolol hydrochloride, 99% (Fisher Scientific Hampton, NH, US), three times a day (8.00 AM; 1.00 PM; 10.00 PM), starting two days before the bacterial challenge (Day -2) until the end of the trial (Day 14). Control animals received vehicle (yogurt and biscuits to facilitate the oral administration of the drug). On the bacterial challenge day (Day 0) a bolus of Escherichia coli (E. coli, 1.4 × 104 CFUs/Kg; strain: ATCC 25,922) was intravenously (i.v.) administered over 15 min at 10.00 AM. Blood samples were collected on the day of the challenge at 1.5, 3, 6, 12 and 24 h after E. coli infusion and then daily (at 10.00 AM). Core body temperature was also measured (Anipill capsule, BodyCAP, Hérouville-Saint-Clair, France; Figure S14). Fourteen days after the immune challenge, animals were anaesthetized as previously described, euthanized by means of i.v. barbiturates overdose (sodium thiopental 40 mg/kg; Penthotal Sodium, MSD Animal Health S.r.l., Rahway, NJ, USA) and subjected to gross post-mortem necroscopy (detailed timeline in Figure S1).
Blood analysis and flow cytometric assay
Blood samples were used for complete blood count (CBC), clinical chemistry, lactatemia, cytokine concentration (MILLIPLEX® Porcine Cytokine/Chemokine Magnetic Bead Panel, Merck Life Science S.r.l., Milan, Italy) and haemocultures (detailed protocol in supplementary methods, page 2).
A flow cytometric phagocytosis assay was performed on blood samples collected before the start of propranolol (or vehicle) treatment (T0), immediately before the bacterial challenge (T1) and then 90 min (T2), 1 and 2 weeks (respectively T3 and T4). Briefly, the blood samples collected with heparin as anticoagulant, were incubated, or not, with fluorescent E.coli Red BioParticles conjugated with a fluorogenic reagent that increases in fluorescence (emission filter 585/26) as the pH decreases in the surrounding environment (e.g. inside lysosomes; pHrodo™ BioParticles™ Conjugates for Phagocytosis and Phagocytosis Kit, Invitrogen™, Waltham, MA, US). The samples were then analysed on the flow cytometer (Attune NxT, Thermofisher, equipped with a 488 nm laser, detailed protocol in supplementary methods, page 2). The white blood cells were identified through their typical localization in a Forward Scatter-Height (FSC-H) vs Side Scatter-Height (SSC-H) dot plot in order to exclude any fragments of the lysed red blood cells. Lymphocytic, granulocytic and monocytic populations were identified and appropriately gated to evaluate their specific physical and fluorescence characteristics. No further counterstaining was performed using conjugated antibodies towards specific cellular surface markers.
Granulocytes gated in negative control samples were analyzed for their light scattering properties: the forward scatter area (FSC-A) values referred to the size of cells were exploited to evaluate the state of activation of neutrophils28. The side scatter high (SSC-H) and side scatter area (SSC-A) referred to the complexity of cells and were used to derive the percentage of disaggregated and aggregated neutrophils29.
We measured the Mean Fluorescence Intensity (MFI) emitted by neutrophils and monocytes after the incubation with fluorescent E.coli Red BioParticles, as a mirror of the amount of BioParticles phagocyted by phagocytes in blood samples (Figure S4 and S15). For each evaluated parameter, a normalization of all timepoints’ value was done by the ratio of each value to the value of the sample collected before the start of pharmacological treatment (T0).
Statistical analysis – animal study
CBC, clinical chemistry, lactatemia and flow cytometry data were analysed using repeated measures ANOVA, with post-hoc comparisons between groups at specific time points using Holm’s correction to the probability level to control for type I error. The assumption of homogeneity of variance was checked using the sphericity test. In case of violation of the assumption, Greenhouse–Geisser’s correction was applied.
Blood bacterial counts, temperature and serum cytokine data were analysed using the non-parametric Kruskal–Wallis test because the frequency distribution of these variables was not normal. When Kruskal–Wallis test was significant, specific time-point comparisons between groups were performed using the Mann–Whitney test.
Data were expressed in the figures as mean ± standard error of the mean (SEM). Missing data were imputed. All analyses were carried out using SPSS software (28.0) The significance level was set to p < 0.05.
Retrospective study on patients
The FINDERS study protocol (#3473) was approved by the Ethics Committee of Romagna Health Authority (CEROM) on December 12, 2022, in accordance with the relevant guidelines. Data were collected in an aggregated and anonymous form. Due to the retrospective nature of the study, the Ethics Committee of Romagna Health Authority (CEROM) waived the need of obtaining informed consent. The datasets generated and/or analysed during the current study are not publicly available due to restrictions imposed by the Ethics Committee’s policy on administrative databases but are available from the corresponding author on reasonable request.
Rehospitalisations with orthopaedic procedure codes ICD-9 CM 76.xx-84.xx occurring within 14 days of discharge from the first hospitalisation were considered as part of the same episode of care and therefore were counted only once.
Only infections not present on index admission were included in the analyses. The infection codes are listed in Table S2 in supplementary materials (Appendix pp 21). Prescriptions for β-blockers were identified by searching the anatomical therapeutic chemical classification system (ATC) codes C07AA (non-selective β-blockers) and C07AB (selective β1-blockers) in the pharmacological dispensing database, that includes retail pharmaceuticals dispensed through a pharmacy and drugs administered or dispensed by the hospital pharmacy at the end of an episode of hospital care. Data extraction was based on the following alternative criteria: i) at least 2 prescriptions on different dates in the 90 days before hospital admission; ii) at least 1 prescription in the 30 days before hospital admission; iii) the sum of the total defined daily dosed in the 90 days before hospital admission was sufficient to encompass the admission date. Other drug prescriptions were classified by methods using the first letter of the ATC nomenclature (see Table S3). For group J “Anti-infectives For Systemic Use” we also determined whether dispensing of the drug had occurred within the 10 days prior to the index hospitalization.
Statistical analysis—patients
Age was summarized as mean ± standard deviation (SD) and compared between groups using the t-test. Categorical data were expressed as absolute and percentage frequencies and compared between groups using χ2 test or Fisher’s exact test. The HAI rate was calculated as the sum of all HAIs divided by the number total number of patients.
Given our focus on non-selective β-blockers, all the primary analyses were conducted using C07AA as the treatment of interest. Conditional logistic regression was used to investigate the association of non-selective β-blockers with HAIs, by matching cases (patients with HAIs) with controls (patients without HAIs) for sex, age group (18–24, 25–29……0.90–94, > 94), and Elixhauser comorbidity index30,31. This index is a comorbidity measure based on International Classification of Diseases (ICD) diagnosis codes, obtained as an unweighted count of comorbid conditions32. Matching was carried out with a ratio 1 case:2 controls using the SPSS procedure CSPLAN.
For descriptive purposes, we also dichotomized the index into three classes based on disease codes from hospitalisations in the previous two years: non-multimorbid (0–1), multimorbid (> = 2), and not available (when information was missing in the hospital discharge records).
Three separate conditional logistic regression models were fitted: one for all infections (matching ratio 1:2; 1 case and 2 controls), one for Gram-negative infections (matching ratio 1:4) and one for Gram-positive infections (matching ratio 1:4). The matching ratio was 1:2 for the all-infection model because it was not possible to identify more than two controls (patients without HAIs) with the same sex, age group and Elixhauser index category for each case (patients with HAIs). For Gram-negative and Gram-positive infections, the matching ratio was 1:4 due to the fewer number of cases. To adjust for the potential effect of antibiotic treatment, a dichotomous variable indicating whether an antibiotic treatment was prescribed in the 10 days prior to the index hospitalization was included in all the models.
To investigate whether the association was specific for non-selective β-blockers, we replicated the above-mentioned analyses using selective β1-blockers as the treatment of interest. This was done under the assumption that the use of the latter does not modify the risk of infection. The analyses were carried out using SPSS 28.0.1.1 and the Stata/SE 17.0 procedure clogit.
link